Saving Us from Ourselves: Harnessing the Power of CAR-T to Combat Autoimmunity
By Michael A. Evans, BSci MSci
Autoimmune disease is a condition in which the body’s immune system mistakenly attacks the body itself.
Thus far, more than 80 different autoimmune diseases have been identified, with some of the most prevalent including rheumatoid arthritis, Crohn’s disease, type 1 diabetes, and multiple sclerosis [1]. Autoimmune disease is one of the top ten causes of death in women under 65 and is the second highest cause of chronic illness in America [2].
These diseases are often life-limiting, debilitating, and, if left untreated, can become life-threatening. Current estimates indicate that roughly 50 million people in the United States are afflicted with an autoimmune disease, representing a substantial economic burden of over 100 billion dollars in annual healthcare costs [3]. Unlike other conditions, such as cancer, there is currently no cure for autoimmune disease, and treatment options are limited.
Overview of autoimmune diseases (courtesy of Indee Labs).
What Are CAR-T Cells?
Since their inception, chimeric antigen receptor T-cell (CAR-T) therapies have undergone extensive study and refinement. These therapies’ potential was realized in 2017 when YescartaTM and KymriahTM were approved for clinical use to treat CD19+ B-cell lymphoma [3]. The lessons learned in developing these therapies have helped define the potential of CAR-T, and it is now clear these cells have clinical relevance far beyond cancer therapy [4,5].
T-cell biology is complex, but two classes of T-cells stand out for their potential in autoimmune CAR-T therapies. Effector T-cells (Teff) defend our body against pathogens and cancer [6]. They do this by directly killing foreign or defective cells and releasing inflammatory factors [18,19].
On the other hand, regulatory T-cells (Treg) are responsible for suppressing an immune response to minimize harm to self. This is achieved by releasing immunosuppressive cytokines and suppressing auto-reactive immune cell populations [15,6,17,19].
When this system functions properly, it allows the body to provide a strong defense against foreign challengers while minimizing the damage to itself. However, an imbalance of one of these subsets can cause or accelerate disease [10,17]. In the case of many autoimmune conditions, the balance tips in favor of Teff cells and leads to the production of inflammation and autoantibodies [14,17].
Autoimmune Diseases On the Rise
The prevalence of autoimmune diseases is on the rise [7,9]. Modern hygiene and disinfection practices have reduced our interactions with the microorganisms critical for teaching our immune system the difference between foreign and self to prevent autoimmune reactions. Because hygiene standards are unlikely to change anytime soon, the global market for autoimmune therapies is predicted to reach $153.32 billion by 2025 [10].
Current treatments for autoimmune diseases are immunomodulating or anti-inflammatory small molecules or proteins designed to help reduce symptoms [11-13]. While these options provide relief for many patients, they have significant limitations, including treatment resistance, continuous administration requirements, and significant side effects [11-15].
For many autoimmune conditions, such as multiple sclerosis, treatments cannot prevent the inevitable disease progression, leaving those in dire need of a better solution [16]. Due to the significant role of T-cells in autoimmunity, CAR-T-based approaches can potentially deliver solutions that can provide long-term patient benefits through symptom reduction, tissue repair, and perhaps a cure [5,6,14,17].
Possible Strategies
Currently, two strategies are the frontrunners for autoimmune CAR-Ts. The first involves a strategy similar to how clinically approved CAR-T therapies function. Teff cells are engineered to possess a chimeric antigen receptor (CAR) with an extracellular component that resembles the self-antigen that the autoantibodies bind to. This allows these CAR-T cells to identify and destroy B-cells producing harmful autoantibodies [6,14].
The approach has been tested in preclinical animal models and has shown tremendous benefit for several autoimmune diseases, including pemphigus vulgaris [19] and lupus [14,22]. It has progressed to a Phase I clinical trial (NCT03030976). Due to its similarity to clinically approved CAR-T therapies, this strategy will likely be the first to market. However, these cells may carry similar risks of cytokine release syndrome and neurotoxicity or therapeutic exhaustion over time which may limit their use [6].
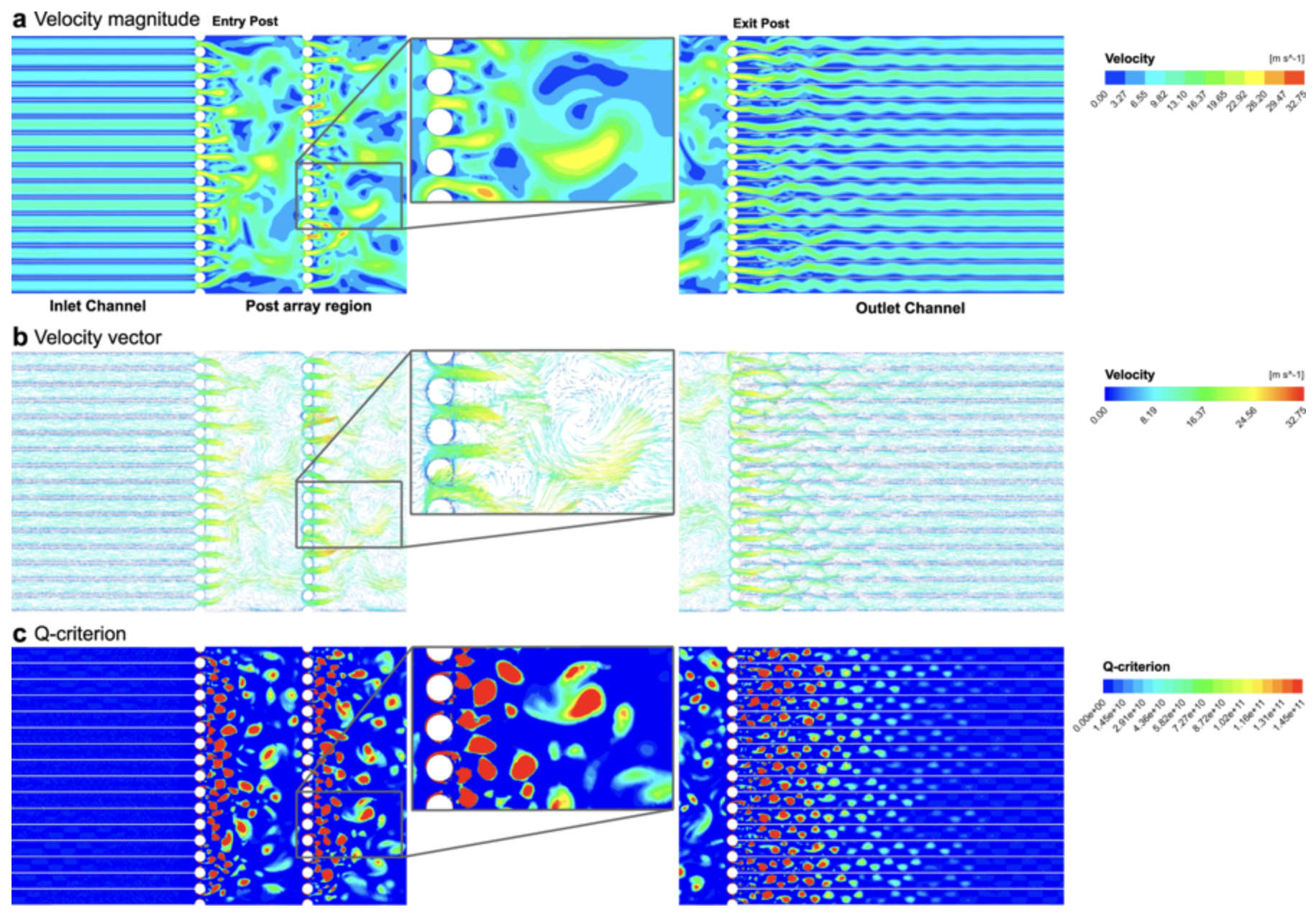
Computational fluid dynamic simulation of in-chip microfluidic vortex shedding [28].
The Second Strategy
The second strategy utilizes Treg cells' immune suppressive nature to control the immune system's activity at the sight of the autoimmune reaction [5]. In this case, CARs are designed to target antigens in the tissue currently under attack [6]. The CAR allows the Treg cells to hone in on the tissue of interest and release a milieu of anti-inflammatory and immunosuppressive factors resulting in a targeted suppressive effect that ameliorates symptoms, promotes long-term tolerance, and stimulates tissue repair [5,6].
This approach has shown significant promise in animal models for colitis [23,24], multiple sclerosis [25], and graft versus host disease [26]. Treg cells are less likely to induce severe side effects seen with Teff cells. However, Treg cells come with some drawbacks [6]. Treg cells only make up 1-2% of the lymphocytes in the blood, making isolation of pure populations in sufficient numbers incredibly challenging [5,6].
In addition, Treg cells can potentially convert to Teff cells in areas of inflammation [6]. If this occurs, it is possible that the CAR receptor on these cells could cause them to attack the tissue, increasing inflammation and accelerating disease progression [6].
This issue can potentially be solved with more advanced strategies, such as including a suicide gene that kills the cell upon losing expression of FOXP3, a regulator of Treg autoimmunity function [6]. Additionally, while the effects of off-target CAR activation for these cells are not as severe as Teff cells, the accumulation of these cells in areas of healthy tissue may help shift a tissue environment in a direction that is supportive of cancer formation and pathogen survival [6].
Challenges Faced and Overcome with Hydropore
One of the biggest hurdles facing autoimmune CAR-T therapy is establishing a production strategy for robust gene transfer and expression in a non-viral manner [27]. Our gene delivery hardware, Hydropore™, provides an excellent solution by employing microfluidic vortex shedding (µVS).
In a recent study published in Nature Scientific Reports, our device was shown to transfect human T-cells rapidly (2 million cells per sec) with a 1 kb mRNA construct with high efficiency (63.6 ± 3.44%) while maintaining high viability (77.3 ± 0.58%) and recovery (88.7 ± 3.21%) of the transfected cells [28]. These results indicate that µVS overcomes many of the critical challenges currently faced by electroporation or viral-based transfection [28].
For Teff cell-based methods, gene transfer mustn't cause premature cell exhaustion, so the cell infusion can impart long-term killing of autoreactive B-cells [29]. Transfection with µVS did not affect growth or critical T-cell activation markers or cytokine excretion, demonstrating that the system did not alter the normal cell function after transfection [28].
Solving the Problem
The potential of Treg cell-based utility is currently hindered by the relative rarity of Treg cells in the blood (1 to 2% of peripheral blood lymphocytes) [5,6]. Indee Labs µVS system will maximize CAR-Treg cells produced for each patient through high yield processing (i.e., cell recovery, cell viability, and modification efficiency). µVS will also permit more rapid expansion of CAR-Treg cells with less perturbation, decreasing over-expansion consequences, such as Treg destabilization [5].
We are excited by the potential µVS holds to help overcome challenges faced in CAR-T manufacturing and believe that it will help accelerate the transition of CAR-T therapies into new areas.
About the Author
Michael A. Evans, BSci MSci is a bioengineering Ph.D. candidate at Harvard University with 7 years of experience in immunology, cell therapies, drug delivery, and organic chemistry. His current research focuses on using macrophages as carriers for nanoparticles to improve their targeting towards inflamed tissues. He is an author of 4 peer reviewed publications with three more in press. He attended Furman University and the University of California Santa Barbara (UCSB), where he received a B.S. and M.S. in chemistry, respectively. He received the 2014 John Sampey Award for the Chemical Sciences and was a 2017 UCSB New Venture Competition Finalist and the People’s Choice Award recipient. Michael will be finishing his PhD in late 2019 and is excited to begin his career in gene-modified cell therapy.
References
https://www.niehs.nih.gov/health/topics/conditions/autoimmune/index.cfm
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1447637/pdf/10983209.pdf
https://www.aarda.org/knowledge-base/many-americans-autoimmune-disease/
Yip, A. & Webster, R. M. The market for chimeric antigen receptor T cell therapies.Nat. Rev. Drug Discov.17, 161–162 (2018).
Bluestone, J. A. & Tang, Q. T reg cells—the next frontier of cell therapy.Science (80-. ).362, 154–155 (2018).
Maldini, C. R., Ellis, G. I. & Riley, J. L. CAR T cells for infection, autoimmunity and allotransplantation.Nat. Rev. Immunol.18, 605–616 (2018).
Cooper, G. S., Bynum, M. L. K. & Somers, E. C. Recent insights in the epidemiology of autoimmune diseases: Improved prevalence estimates and understanding of clustering of diseases.J. Autoimmun.33, 197–207 (2009).
Okada, H., Kuhn, C., Feillet, H. & Bach, J. F. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: An update.Clin. Exp. Immunol.160, 1–9 (2010).
Bach, J. F. The hygiene hypothesis in autoimmunity: The role of pathogens and commensals.Nat. Rev. Immunol.18, 105–120 (2018).
Correa, D. Global Autoimmune Disease Therapeutics Market to Garner $ 153.32 Billion by 2025 at 4.2 % CAGR,Says Allied Market Research.Global News Wire(2019). Available at: https://www.globenewswire.com/news-release/2019/03/13/1752518/0/en/Global-Autoimmune-Disease-Therapeutics-Market-to-Garner-153-32-Billion-by-2025-at-4-2-CAGR-Says-Allied-Market-Research.html. (Accessed: 5th December 2019)
Chan, A. C. & Carter, P. J. Therapeutic antibodies for autoimmunity and inflammation.Nat. Rev. Immunol.10, 301–316 (2010).
Olivera, P., Danese, S. & Peyrin-biroulet, L. Next generation of small molecules in inflammatory bowel disease.Gut66, 199–209 (2017).
Hall, B. M., Hodgkinson, S. J. & Quin, J. Corticosteroids in autoimmune diseases.Australian Prescriber(1999).
Tahir, A. Is Chimeric Antigen Receptor T-cell Therapy the Future of Autoimmunity Management?Cureus10, 3–5 (2018).
Pichler, W. J. & Campi, P.Adverse side effects to biological agents.Drug Hypersensitivity(2007).
Wingerchuk, D. M. & Weinshenker, B. G. Disease modifying therapies for relapsing multiple sclerosis.BMJ354, (2016).
Sakaguchi, S., Yamaguchi, T., Nomura, T. & Ono, M. Regulatory T cells and immune tolerance.Cell133, 775–787 (2008).
Janeway, C. A., Travers, P., Walport, M. & Shlomchik, M. J.Immunobiology: The Immune System in Health and Disease.Garland Science(2001).
Pennock, N. D.et al.T cell responses: naïve to memory and everything in between.Adv. Physiol. Educ.37, 273–283 (2013).
Ward-Hartstonge, K. A. & Kemp, R. A. Regulatory T-cell heterogeneity and the cancer immune response.Clin. Transl. Immunol.6, e154 (2017).
Ellebrecht, C. T.et al.Reengineering chimeric antigen receptor T cells for targeted therapy of autoimmune disease.Science (80-. ).353, 179–184 (2016).
Leick, M. B. & Maus, M. V. CAR-T cells beyond CD19, UnCAR-Ted territory.Am. J. Hematol.34–41 (2019). doi:10.1002/ajh.25398
Elinav, E., Waks, T. & Eshhar, Z. Redirection of Regulatory T Cells With Predetermined Specificity for the Treatment of Experimental Colitis in Mice.Gastroenterology134, 2014–2024 (2008).
Blat, D., Zigmond, E., Alteber, Z., Waks, T. & Eshhar, Z. Suppression of murine colitis and its associated cancer by carcinoembryonic antigen-specific regulatory T cells.Mol. Ther.22, 1018–1028 (2014).
Fransson, M.et al.CAR/FoxP3-engineered T regulatory cells target the CNS and suppress EAE upon intranasal delivery.J. Neuroinflammation9, 1 (2012).
Broady, R.et al.Alloantigen-specific regulatory T cells generated with a chimeric antigen receptor.J. Clin. Invest.126, 1413–1424 (2016).
Piscopo, N. J.et al.Bioengineering Solutions for Manufacturing Challenges in CAR T Cells.Biotechnol. J.13, 1–10 (2018).
Jarrell, J. A.et al.Intracellular delivery of mRNA to human primary T cells with microfluidic vortex shedding.Sci. Rep.9, 1–11 (2019).
Zhang, M.et al.The impact of Nucleofection® on the activation state of primary human CD4 T cells.J. Immunol. Methods408, 123–131 (2014).